Etching the PCB
The APL has a container of etchant, copper chloride (CuCl2) mixed with hydrochloric acid (HCl), that should be used for etching the PCB. These chemicals are dangerous and should be handled with care.
Hydrochloric acid (HCl) is very acidic and should not touch your skin and copper chloride is extremely harmful to water treatment plants, so DO NOT pout this solution down the drain (see the warning in the After Etching section). Fortunately, there should be no need to dispose of any etchant because the etchant is reusable after the etching process (see how in the Technical Information section).
Also note that the fumes from the etchant are dangerous, thus the etchant should only be opened and used under a fume hood (Dr.Boggs has access to one in CAMCOR).
Contents
Testing the CuCl2
Before using the CuCl2 you want to make sure that the etchant has the correct pH balance and the correct specific gravity.
The ideal pH of the solution is < 1 and the ideal specific gravity is between 1.2393 and 1.3303.
Testing the pH
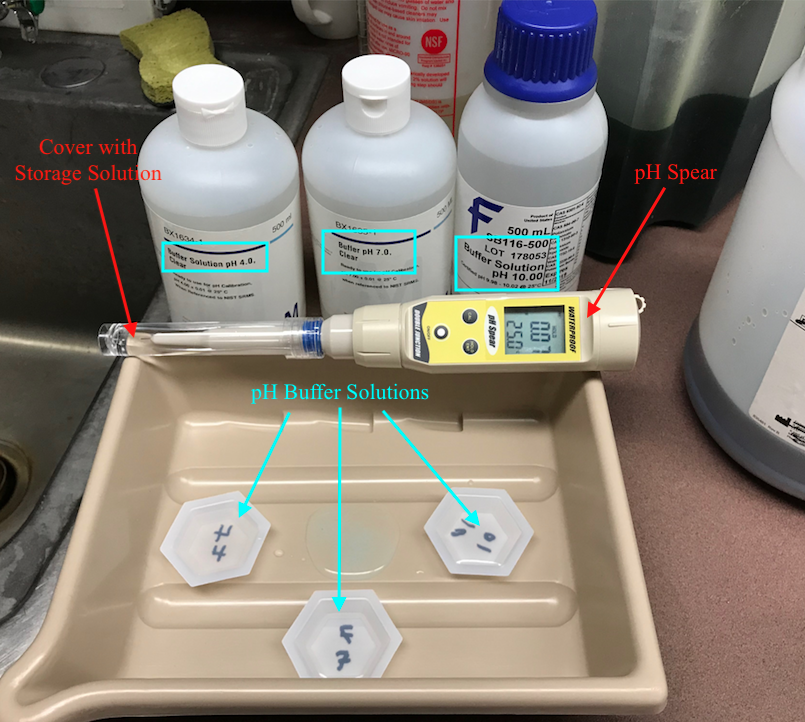
To test the pH you'll want to use a pH meter. In the APL we have one called a pH spear. This can be calibrated and then used to test the pH of the etchant. Calibration should be done once a week.
To calibrate the pH meter you will need 3 clear, liquid solutions called pH buffers. Each pH buffer has a precisely determined pH that can be used to calibrate the pH meter. First, put on eye protection and gloves. Then, set up the three buffer solutions in three labeled trays like the picture below (make sure to write on the bottom side of the trays to avoid changing the pH of the solution in the tray) and place the three small trays in a large tray to contain spillage or dripping. Also, make sure the solutions are about room temperature and then proceed to calibration.
When you are ready to calibrate the pH meter, turn on the meter by pressing the ON/OFF button. Next, carefully remove the storage cap and solution from the tip of the pH meter. Make sure the storage solution does not spill and place the cap in a secure location for later. Rinse the tip of the pH spear with deionized water to remove the storage solution from the tip. Be sure to rinse the pH spear with deionized water between every submergence in a different liquid to wash away the previous liquid. Next, press the CAL button to begin calibration. Submerge the tip of the pH spear 2cm - 3cm below the surface of the 4 pH buffer solution for 2 minutes (this allows the electrode in the tip to stabilize). The screen will display two numbers, the top number is the reading for the current pH and the bottom number is the pH the meter is being calibrated to. Ideally, for the pH 4 buffer solution the top number will gradually drop or rise to 4.01 while the bottom number remains at 4.01 (the bottom number will only ever display 4.01, 7.00, or 10.01 which correspond to the different buffer solutions).
After 2 minutes in the solution, without removing the tip from the solution, press the HOLD/ENT button, this will save the current calibration. Rinse the tip with deionized water and submerge the tip in the pH 7 buffer solution. Once again press enter after 2 minutes and rinse the tip. Finally repeat this process for the pH 10 solution and rinse the tip with deionized water.
Now that the pH meter is calibrated, dip the pH meter 2cm - 3cm into the etchant and record the pH. At the time of writing this the pH of the solution is 0.07, which is well below 1 and thus adequate. When you remove the pH Spear from the etchant the tip will have a small amount of bright green etchant on it, because the etchant is caustic and should not go down the drain (see warnings below) you do not want to immediately rinse the tip with deionized water. First, while wearing gloves, gently dry the green liquid from the tip with kimtech wipes. This will absorb the excess liquid and you can then rinse the tip with deionized water.
If you are finished with pH meter return the pH spear into the storage cap with the storage solution and appropriately dispose of your excess chemicals (do not just pour them down the drain). Also, after adjusting the pH, record the date and pH on the side of the container.
If any of these instructions were unclear please refer to the pH Spear manual, which should be with the pH Spear.
Decreasing the pH
If the pH is above 1, you can decrease the pH by adding more HCl to the etchant. Handling pure HCl is dangerous and should be done with extreme caution. Ask professor Boggs for help with this.
Testing the Specific Gravity
To test the specific gravity you will need a hydrometer, again the ideal range is between 1.24 and 1.33. This will determine the relative density of the etchant. Using the hydrometer just requires you to place the weighted end down in the liquid, the hydrometer will then float (make sure there is enough liquid in your container to ensure the hydrometer will not touch the bottom of your container). The level at which the hydrometer floats in the liquid indicates the specific gravity, thus after a few seconds the hydrometer will stabilize and you can read the specific gravity from where the markings on the inside of the tube match with the top surface of the liquid.
If the specific gravity is too high you need add water. The specific gravity should not be too low because the etching process increases the specific gravity, although if too much water is added and brings the specific gravity down below 1.24 you can still perform suboptimal etching that will increase the specific gravity in the process.
Lastly, after adjusting the specific gravity, date and record your measurement on table tapped to the side of the container.
Using the CuCl2
Before interacting with the chemicals, make sure you have latex gloves, safety glasses, and a lab coat on.
Etching with the Aggregation Tank
Before you start the etching process you will want to set up the thermometer in the aggregation tank, turn on the heating pillar by plugging it in, start the bubbler by plugging it in, and set up a tray of water/sodium bicarbonate mix (this mix should contain very roughly 1 tablespoon of sodium bicarbonate and 500 mL of water). The sodium bicarbonate will be used to neutralize the HCl in the CuCl2 and halt the etching process after you are finished. While increasing the temperature increases etching speed, it also boils off more HCl (which has a very low boiling point). I recommend bringing the CuCl2 solution up to 32 degrees Celsius and then unplugging the heater. The solution should roughly maintain this temperature for the duration of the etching process without being plugged back in.
The aggregator with the CuCl2 should look like this when set up, but make sure this is done under a fume hood (the HCl fumes are toxic and cause rust). Also make sure you have your sodium bircarbonate solution set up near by.
Once you're all set up and the etchant is up to temperature you can attach you're PCB to a clip and submerge it in the liquid. You're goal is to remove all the copper from the areas without the green photoresist. You'll need to continue the etching process as long as it takes to do this, although my PCB took about 13.5 minutes so you can use this as a rough estimate. This time will not be the same for you and you must check the board at least every 3-5 minutes, if the board is left unattended for too long even the copper under the photoresist will be removed and you will just be left with a piece of fibreglass. As the copper is removed you will begin to see the fibreglass board. You're etching process is complete when all that is left on the board is the green photoresist (which protects the copper layer below it) and fibreglass. There should be no visible copper.
Etching without the Aggregation Tank
Before you start etching make sure you set up a tray of sodium bicarbonate and water to neutralize the acids after etching. The action is slow at our room temp (22C) and may require as long as 30 minutes of agitation. Agitation *must* be continuous (and currently, by hand as we lack a bubbler). The etching process occurs at the surface and not in the volume, thus it is a diffusion limited process - agitation is *essential* or the metal simply forms a depletion zone adjacent to itself devoid of etching ions. Etching is complete once close inspections shows NO remaining exposed copper.
Note that the board cannot be left to etch unattended even with automatic agitation - the acid etches across as it cuts down (at a fairly small angle), but if left indefinitely it will eat *all* the copper off the board.
After Etching
Once etching is complete, remove the board from the etchant with a gloved hand. Try to shake as much etchant as possible back off into the tank before putting the board into a prepared small tray of sodium bicarbonate solution. This will immediately halt the etch action, and remove all remaining copper ions from the etchant solution that remained to the board. You will see light blue particles form in the sodium bicarbonate solution when it contacts the etchant.Gentle brushing will take any copper bicarbonate (the light blue particles) off the board.
Once the board is clean, we want to remove the remaining photoresist from the copper traces. Pour a very small amount of acetone over the board and you should see the green layer disappear to reveal your copper traces. This step is necessary so you will be able to solder components to the copper of the board.
Etchant Warning
Make sure none of the etchant goes down the drain because putting copper ions down the drain is very illegal:
https://nature.nps.gov/water/ecencyclopedia/assets/contaminant-pdfs/copper.pdf
Copper ion concentrations measured in single-digit parts per million are dangerous to aquatic life, and the required dilutions for copper ion contaminated water to go down the drain are concomitant: Our tank of etchant would contaminate several olympic swimming pools of water beyond legal release limits.
Under no circumstances allow any amount of green etchant solution to go down the drain! If any does, dump the sodium bicarbonate in immediately, chase it with a torrent of water, and pour additional bicarbonate in the sink straight from the box to 'kill it' before it enters the wastewater stream. If substantial spillage (more than a thimble full) occurs, get Dr. Boggs and call EHS immediately. A large release of copper ions down the drain threatens the entire city's wastewater treatment plant.
Technical Information
The Acopper chloride etchant functions via the following reactions. The obvious attraction of this process is that it produces no waste stream: The result of etching copper is more etchant! Be very careful with the etchant as HCL is extremely acidic, also see the warnings below about the proper way to dispose of the etchant (do not pour it down the drain).
Cu(s) + CuCl2 -> 2CuCl
CuCl + chlorinating oxidation -> CuCl2
The tank contains a not insignificant amount of hydrochloric acid (the unmistakeable smell of which is detectable when it is open). However, the HCl is not free: It is almost entirely bound up in complexes with the copper chloride. Transition metal salts are known for being extremely colorful, and copper is no exception.
Dilute solutions of copper chloride in water are a serene blue (the copper ion coordinates with 6 waters). Intermediate concentrations are yellowish, and the addition of hydrochloric acid to a concentrated solution results in the copper ions forming a complex with two waters and four chloride ions (CuCl_4^{-2} \cdot 2H_2O), and it is this complex which gives our etchant its beautiful and almost hypnotically intense green hue.
The second reaction above is a summary of what is actually a rather complicated series of ion-complex reactions. The first reaction proceeds rapidly, while the second is the rate limiting factor (the insertion of a powerful oxidizer, like hydrogen peroxide, to the solution can speed this up).
This can be observed if the board is withdrawn entirely from the etchant solution to examine: Immediately, brownish liquid can be seen draining off of all exposed copper areas - these drops are etchant in which all available acid has been used up and copper (II) has saturated the solution with brown copper (I) ions.
This underscores why turbulent action is essential to the etching process: Like photographic development, etching is a surface reaction whose components come from a bulk fluid, and the bulk must be turbulently washed over the surface to continuously bring "new" etchant, unladen with copper I ions, within diffusion range of the surface.
If the CuCl2 becomes over saturated during the etching process the addition of hydrogen peroxide will restore the copper chloride to its original state.